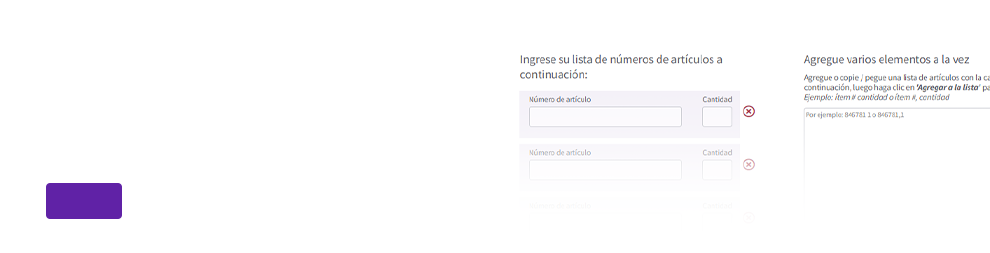
Por favor, introduzca un texto válido.
Ha aceptado un presupuesto y actualmente está disponible en su carrito. Complete el procesamiento del pedido para agregar un artículo al carrito.
Welcome to GE HealthCare
Customer agrees that products purchased from the GE HealthCare Service Shop will only be used in operating and servicing medical equipment at a healthcare facility located in the United States in which it was intended, and will not be otherwise used for manufacturing, resale or other distribution.
Este formulario completará automáticamente la información de la cuenta para los usuarios registrados.
¿Cómo quiere proceder?
Proceed as Guest LoginPor favor, introduzca la dirección de correo electrónico
Recuérdame
Olvidó su contraseña?Preguntas sobre su cuenta? Contáctenos.